Autoimmune Hemolytic Anemias (AIHA)
Autoimmune hemolytic anemia is a group of diseases
characterized by an impaired immune system, resulting in the production of
autoantibodies that treat red blood cells as heterologous and attack them.
Some people with asymptomatic, and
other people are tired, short of breath, and pale.
• Usually Immune tolerance prevents the formation of autoantibodies
– Autoimmune diseases occur due to:
▪ Genetic predisposition
▪ Exposure to infectious agents (molecular mimicry)
▪ Defects in the regulation of immune tolerance
– Subcategories
▪ Warm autoantibodies
▪ Cold autoantibodies
–Further categorized as primary versus secondary AIHA
Warm AIHA
• Accounts for ~ 70% of cases AIHA
–Optimal reactivity at 37°C
– Usually IgG (rarely IgM, IgA)
– Most Abs react with "Rh protein" complex
▪ Not react with Rh null or Rh deleted cells
▪ Sometimes have single specificity within the Rh system (e.g., anti-e).
• Accounts for ~ 70% of cases AIHA
–Most hemolysis is extravascular through splenic macrophages.
• Idiopathic WAIHA
– Accounts for about 60% of cases of warm AIHA
– Acute idiopathic WAIHA
▪ Severe anemia
▪ Developing over two to three days
▪ Hemolysis is self-limited
▪ Several weeks to several years’ duration
– Chronic idiopathic WAIHS
▪ Sustained Hemolysis.
• Secondary WAIHA associated with:
– Lymphoproliferative disease: CLL, HD.
– Neoplastic diseases
– Autoimmune disorders
▪ SLE, RA, Crohn's disease.
– Certain viral and bacterial infections
– Certain Vaccines
• Clinical findings
– Can occur at any age
▪ Incidence increase after age 40
– Common presentation
▪ Symptoms of anemia
– Secondary AIHA
▪ Signs and symptoms of the underlying disorder
– Mild to moderate splenomegaly is more than 50% of patients, hepatomegaly in one-third of patients.
• Laboratory findings
–Immune-mediated hemolysis
▪ Positive DAT
▪ Autoantibody in the serum
▪ Presence of spherocytes
– Peripheral blood
▪ Moderate to severe normocytic/normochromic anemia
▪ Reticulocytosis
▪ Polychromasia, NRBCs, spherocytes, schistocytes
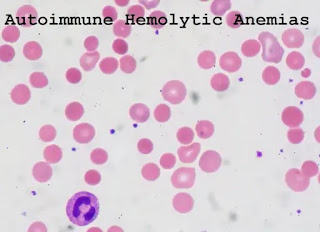
Blood film from a patient with warm autoimmune hemolytic anemia (WAIHA).
The obvious anisocytosis is due to the presence of spherocytes and large polychromatophilic erythrocytes.
those nucleated cells are erythroblasts (Wright stain, 1000× original magnification).
– Bone marrow
▪ Erythroid hyperplasia, erythrophagocytosis
– Other lab tests
▪ Positive DAT
– Positive polyspecific AHG and anti-IgG monospecific AHG
–30% + anti-C3
– Differential diagnosis from HS
▪ Positive DAT
▪ Autohemolysis test not corrected by glucose
▪ The non-homogeneous population of spherocytes
Laboratory findings associated with WAIHA
Common findings:
- Positive DAT
- Normocytic normochromic anemia
- Increased reticulocytes
- Spherocytes and other erythrocyte abnormalities
- Presence of autoantibodies in the serum
- Increased serum bilirubin (total and unconjugated)
- Decreased serum haptoglobin
- Elevated lactate dehydrogenase (LDH)
- Positive antibody screen include all cells even auto control.
- Incompatible cross matches with all donors
Other laboratory findings associated with hemolysis in WAIHA:
- Increased osmotic fragility
- Increased urine and fecal urobilinogen
- Hemoglobinemia, hemoglobinuria, methemoglobinemia, hemosiderinuria.
• Therapy
– Self-limited hemolytic disorders
▪ Do not require transfusions.
▪ Immunosuppressive drugs, cytotoxic drugs
▪ Splenectomy
▪ Rituximab
▪ IVIG—high dose intravenous immunoglobulin
▪ Plasma exchange and plasmapheresis
Cold AIHA
• Accounts for ~ 16–30% of cases of AIHA
– Optimal reactivity < 37°C
– Usually IgM, with complement activation
▪ Rarely IgA or IgG
▪ Highest Abs react with I/i Ags or Pr Ags
• Accounts for ~ 16–30% of cases of AIHA
– Severity of disease
▪ Associated with the thermal range of the Ab
▪ Most hemolysis due to complement-mediated lysis
• Idiopathic or secondary
– Idiopathic CAS (cold agglutinin syndrome)
▪ Usually chronic, occurring after age 50)
▪ Ab involved is usually
–Monoclonal IgM/kappa with autoanti-I specificity
– Secondary CAS
▪ Associated with infectious disease
–Usually acute, self-limiting
–Polyclonal autoAbs with specificity for Ii antigens
• anti-I—M. pneumoniae
• anti-I—Infectious mononucleosis
• anti-Pr—varicella, rubella
▪ Associated with lymphoproliferative disorders
–Usually chronic, found in older individuals
–Monoclonal IgMκ Ab
Autoimmune hemolytic anemia caused by cold-reacting antibodies
cold agglutinin syndrome:
- Primary (Idiopathic)
- Secondary:
▪ Viral and bacterial infections
▪ Mycoplasma pneumonia infections, infectious mononucleosis.
▪ Lymphoproliferative diseases.
Paroxysmal cold hemoglobinuria
- Primary (Idiopathic)
- Secondary:
▪ Viral infections
▪ Tertiary syphilis
• Clinical findings
– Chronic or episodic hemolytic anemia
– RBC agglutination
▪ Sludging of blood flow within capillaries
– Vascular changes
▪ Acrocyanosis
▪ Raynaud's phenomenon
–Pain with color change patterns in skin
– Hemoglobinuria and splenomegaly
• Laboratory findings
– Automated blood counts
▪ Falsely decreased RBC
▪ Falsely increased MCV
▪ Must warm blood and diluting reagents
– Mild to moderate anemia
▪ Normocytic/normochromic
▪ Polychromasia, spherocytes, clumps of RBCs, NRBCs, erythrophagocytosis
– Bone marrow
▪ Normoblastic hyperplasia
Criteria for clinical diagnosis of cold agglutinin syndrome
- Clinical history
▪ Acrocyanosis
▪ Hemoglobinuria on exposure to cold
- Laboratory findings
▪ Serological:
- DAT: with polyspecific AHG Positive, with anti-IgG Negative, with anti-C3 positive.
- IAT: Antibody showing characteristic reaction at,25oC.
- Cold agglutinin titer >1000 at 4oC.
▪ False increase in MCV, MCH, and MCHC.
▪ False increase in erythrocyte count.
▪ Normocytic normochromic anemia
▪ Reticulocytosis
▪ Presence of Spherocytes, agglutinated RBCs, rouleaux, nucleated RBCs on the blood film.
▪ Increased bilirubin (total and unconjugated)
▪ Decreased Haptoglobin
▪ Hemoglobinemia, hemoglobinuria in acute hemolysis.
▪ Hemosiderinuria in chronic hemolysis.
Cold autoimmune hemolytic anemia from patients with chronic lymphocytic leukemia. Some of the erythrocytes are in small clumps. Spherocytes are also present in the peripheral blood.
– Differential diagnosis
▪ Benign cold autoagglutinins vs pathologic cold agglutinins
–Pathologic cold agglutinins
• Positive DAT for polyspecific AHG and monospecific anticomplement antiserum
• Cold agglutinin test—agglutinates RBCs at 0–20°C in saline, reversible at 37°C
• Titer usually > 1:1000 (normal 1:64)
Comparison of the characteristics of pathological cold agglutinins found in CAS and benign cold agglutinins in normal people
|
|
Pathologic agglutinins
|
Benign agglutinins
|
|
Antibody class
|
IgM
|
IgM
|
|
Antibody specificity
|
Usually anti-I but in secondary CAS can be anti-i
|
anti-I
|
|
Antibody clonality
|
Monoclonal in idiopathic type and secondary type due to
lymphoproliferative diseases; polyclonal in secondary type due to infectious
diseases
|
Polyclonal
|
|
Thermal Amplitude
|
0-30oC
|
0-4oC
|
|
Agglutination at room temperature
|
Significant
|
Not present
|
|
Titer
|
Usually > 1:1000
|
< 1.64
|
|
DAT
|
Positive with polyspecific AHG and monospecific
anticomplement
|
Negative
|
• Therapy
– Keeping extremities warm is most effective
– Secondary to lymphoproliferative disorder
▪ Chemotherapy
– Plasma exchange for acute hemolytic episodes
▪ Effective for a short time
Paroxysmal Cold Hemoglobinuria (PCH)
• Rare autoimmune hemolytic disorder
– Can occur at any age
– Massive intermittent acute hemolysis and hemoglobinuria
– Accounts for 30–40% of all AIHA in children
▪ Most frequent < age 5
▪ Associated with viral and bacterial infections
• Rare autoimmune hemolytic disorder
– Usually transient disorder
▪ Resolves spontaneously
– Transfusions may be needed in severe cases.
• Pathophysiology
– Bi-phasic complement-fixing IgG antibody
▪ Donath Landsteiner antibody
▪ Binds RBCs at low temps (< 20°C), activates complement
– Bi-phasic complement-fixing IgG antibody
▪ Upon warming to 37°
–Ab detaches
–RBC lysed by complement activation through C9 (MAC)
▪ Usual reactivity—autoanti-P antibody
• Clinical findings
– Hemoglobinuria most common clinical symptom
– Jaundice, pallor, hepatosplenomegaly
– Raynaud's phenomenon can occur
• Laboratory findings
– Anemia depends on the frequency and severity of attacks.
– Hb dropped sharply to 5 g/dL.
– Hemoglobinemia, methemalbuminemia, hemoglobinuria
– Neutropenia, neutrophil shift to left
– Reticulocytopenia, spherocytes
– ↑ serum bilirubin, BUN, LD
– ↓ serum complement, haptoglobin
– Erythrophagocytosis
– DAT usually negative for antibodies
– DAT weakly + for the complement
– IAT + if performed in cold
– Donath-Landsteiner antibodies
▪ Present in low titers < 1:32
▪ Verified by D-L test
Donath-Landsteiner (D-L) test to detect the presence of D-L antibodies
|
Patient’s whole
blood
|
Control
|
Test
|
|
Incubate for 30 min at
|
37oC
|
4oC
|
|
Incubate for 30 min at
|
37oC
|
37oC
|
|
Centrifuge: observe plasma for the presence of
hemolysis
|
|
|
Interpretation
|
|
D-L antibodies present
|
No hemolysis
|
Hemolysis
|
|
No D-L antibodies present
|
No hemolysis
|
No hemolysis
|
• Therapy
– PCH terminates with recovery from infection.
– Transfusion if hemolysis is severe
– Plasmapheresis if hemolysis persists
– Rituximab
– Avoid exposure to cold
Comparison between cold agglutinin syndrome (CAS) and paroxysmal cold hemoglobinuria (PCH)
|
|
CAS
|
PCH
|
|
Patient
|
Usually adult > 50 years of age
|
Usually children after viral infection
|
|
Clinical findings
|
Acrocyanosis
|
Chills, fever, hemoglobinuria
|
|
DAT
|
Positive with polyspecific AHG and monospecific C3
|
Positive with polyspecific AHG and monospecific C3
|
|
(D-L) test
|
Negative
|
Positive
|
|
Antibody class
|
IgM
|
Biphasic IgG (D-L)
|
|
Antibody specificity
|
Anti-I
|
Anti-P
|
|
Thermal Amplitude of antibody
|
Up to 30oC
|
Under 20oC
|
|
Hemolysis
|
Chronic extra/intravascular
|
Acute intravascular
|
|
Therapy
|
Avoid the cold
|
Supportive; treatment of underlying illness
|
Mixed-Type AIHA
• Because of the presence of warm-reacting IgG autoAb and cold-reacting IgM autoAb
– Both have raised titer and increased thermal amplitude
– 50% of cases are idiopathic.
– The rest are related to lymphoproliferative diseases and autoimmune diseases (e.g., SLE), or HIV
• Because of the presence of warm-reacting IgG autoAb and cold-reacting IgM autoAb
– A combination of intravascular (IgM) and extravascular (IgG) hemolysis.
–Most patients respond to corticosteroids without transfusions.



Comments
Post a Comment